I really enjoyed going to this year’s MERCURY conference. In fact, a few of the speakers were very inspirational, as were some posters. The speakers that I most enjoyed were Kate Holloway and Chris Wilmer. Kate Holloway talked about drug design to combat HIV and HCV and I was really intrigued by the details and difficulties in this process. Chris Wilmer discussed MOF’s and how they can be used to model methane consumption. I think I liked these presentations the most, both because the speakers were very good and also because I could clearly see the applications of their research in our lives. I also really enjoyed the poster session. My presentation went well, except I was asked a few questions I was uncertain about and Chris Cramer didn’t come to talk to me 🙁 . However, I enjoyed looking at other posters more than presenting my own. There were three posters that I enjoyed the most. First, a student researched nano-aggregates and measured their speed of aggregation to be able to find a solution to clogging when these nano-aggregates interact with waxes in pipes. Her research seemed very simple on the poster, but the work was quite fascinating. Second, a student experimented with different halogens and leaving groups to see if he could make a bi-molecular reaction become a concerted reaction. I don’t think I realized how many different directions molecular dynamics could go; including organic chemistry. Lastly, two girls presented their poster about enzyme inhibitors. I can’t quite remember the entire purpose but I know that they were working with inhibitors, derivatives of some molecule (which included tryptophan), and dopamine. They measured interaction energies between the derivatives and dopamine to find the best one, with two different conformations.
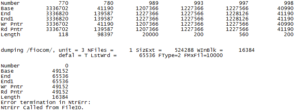
I was disappointed that I wasn’t able to get any suggestions about Gaussian’s confusion problem from any of the quantum professors or speakers there who are familiar with the program. I would have liked to been able to discuss more with them the problems that we have been having. Overall, I enjoyed seeing the subjects that other students were researching with computational chemistry. Also, the conference made me sad that I am not taking a science class next semester. . .